Why test for homologous recombination deficiency (HRD) in advanced ovarian cancer?
Ovarian cancer is the most common cause of death among gynecologic cancers.1
Newly diagnosed advanced ovarian cancer is treated with curative intent using cytoreductive surgery and systemic therapy. The first line is likely to be the best setting for treatments to help prevent recurrence.2,3
HRD testing is critical to inform treatment eligibility in patients with advanced ovarian cancer.4
Many patients with ovarian cancer are not tested for BRCA mutations or HRD5
- In a retrospective study of biomarker testing in 1002 patients with epithelial ovarian cancer drawn from community health systems in the US (2018–2020), 74.7% (748/1002) underwent testing.5
Upfront testing for HRD, prior to initiation of primary therapy, can identify patients who are eligible for LYNPARZA plus bevacizumab maintenance therapy.4
HRD in Ovarian Cancer
The homologous recombination repair (HRR) pathway repairs double-strand breaks in DNA.6
Defects in the HRR pathway lead to HRD, the inability to accurately repair double-strand DNA breaks.6
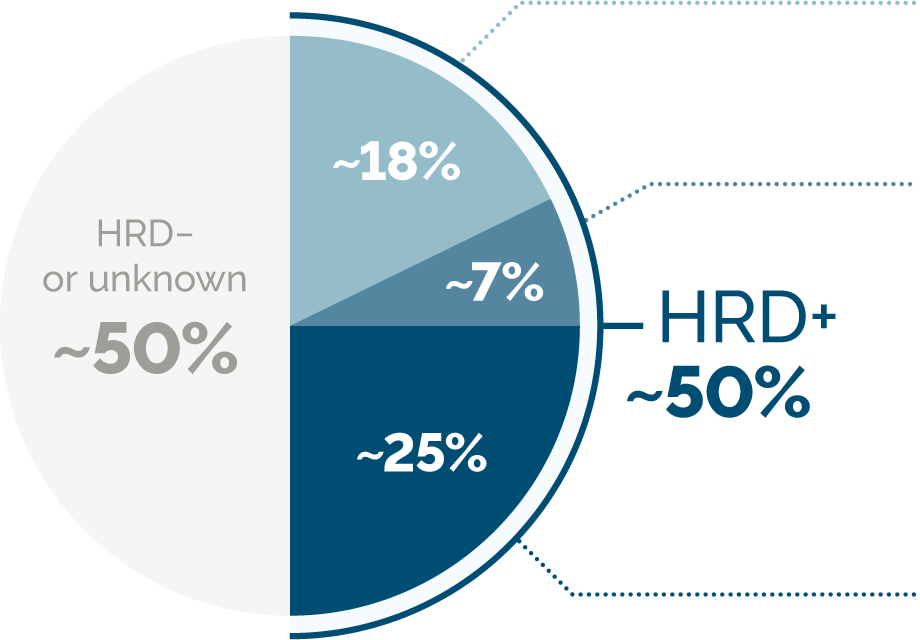
About half of high-grade serous ovarian cancers exhibit HRD.7
Germline testing is conducted on blood samples, whereas somatic and HRD testings are conducted on samples of tumor tissue, and ideally at diagnosis.9,10,12
| Test type | Tumor | Germline |
|---|---|---|
| Test typeSample | Tumor Fresh/frozen sample or archived FFPE specimen10 |
Germline Blood10 |
| Test typeClinical relevance | TumorDetermines total mutation status (somatic + germline)10 May be used to assess genomic instability11 May inform magnitude of benefit of PARP inhibitor therapy13 Tumor testing for BRCA mutations and HRD including genomic instability tests identifies more patients than testing for germline BRCA mutations alone.7,8 |
GermlineMay have familial implications14 Informs maintenance therapy13 |
| Test typeLimitations | TumorSamples can contain both malignant and normal cells; low cancer cell content can affect results10,11 Does not distinguish between somatic and germline mutations10 |
GermlineDoes not identify somatic mutations10 |
FFPE, formalin-fixed, paraffin-embedded; PARP, poly (ADP ribose) polymerase.
HRD/BRCA Tests Available in Ovarian Cancer
HRD and BRCA companion diagnostic (CDx) tests available for advanced ovarian cancer are shown in the Table.
| Test (company)* | Sample type | gBRCA test† | Tumor BRCA test‡ | HRD test | Turnaround time | Approved as CDx in advanced ovarian cancer for? |
|---|---|---|---|---|---|---|
| Test(company)*BRACAnalysis CDx®(Myriad Genetics) | Tumor Whole blood |
gBRCA test† |
Tumor BRCA test‡ | HRD test | Turnaround time<14 days | Approved as CDx in advanced ovarian cancer for?
|
| Test(company)*MyChoice® CDx (Myriad Genetics) | Tumor FFPE tumor specimen |
gBRCA test† | Tumor BRCA test‡ |
HRD test |
Turnaround time≤14 days | Approved as CDx in advanced ovarian cancer for?
|
| Test(company)*FoundationOne® CDx (Foundation Medicine) | Tumor FFPE tumor specimen |
gBRCA test† | Tumor BRCA test‡ |
HRD test |
Turnaround time≤12 days | Approved as CDx in advanced ovarian cancer for?
|
BRCAm, BRCA mutation; CDx, companion diagnostic; FFPE, formalin-fixed, paraffin-embedded; gBRCA, germline BRCA; HRD, homologous recombination deficiency.
*CDx tests only; not an exhaustive list of available tests.
†Detects germline BRCA mutations only; does not detect somatic mutations or HRD.
‡Tumor BRCA tests do not distinguish between germline and somatic BRCAm.
Maximize identification of patients eligible for LYNPARZA plus bevacizumab by using Myriad MyChoice, an FDA-approved HRD test that measures both BRCA mutations and genomic instability.4,17

Dr. Ramez Eskander explains the importance of germline and tumor testing.
“I test everybody. I do it concurrently.”

Dr John Chan describes the take home message about genetic testing in women with ovarian cancer.
“All women should be tested.”
References
- American Cancer Society (ACS). Cancer Facts & Figures 2025. Atlanta, GA; 2025. Accessed August 28, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2025/2025-cancer-facts-and-figures-acs.pdf
- Ray-Coquard I, Leary A, Pignata S, et al. Olaparib plus bevacizumab first-line maintenance in ovarian cancer: final overall survival results from the PAOLA-1/ENGOT-ov25 trial. Ann Oncol. 2023;34(8):681-692.
- Lorusso D, Mouret-Reynier M-A, Harter P, et al. Updated progression-free survival and final overall survival with maintenance olaparib plus bevacizumab according to clinical risk in patients with newly diagnosed advanced ovarian cancer in the phase III PAOLA-1/ENGOT-OV25 trial. Int J Gynecol Cancer. 2024;34(4):550-558.
- LYNPARZA® (olaparib) [prescribing information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025.
- Matsuno RK, Williams AFO, Sweetnam C, et al. Patterns of homologous repair deficiency and BRCA1/2 testing of ovarian and breast cancers: A real-world study of patients in community health settings in the United States. JCO Oncol Advances. 2025;(2).
- O’Connor MJ. Targeting the DNA damage response in cancer. Mol Cell. 2015;60(4):547-560.
- Konstantinopoulos PA, Ceccaldi R, Shapiro GI, D’Andrea AD. Homologous recombination deficiency: exploiting the fundamental vulnerability of ovarian cancer. Cancer Discov. 2015;5(11):1137-1154.
- Pennington KP, Walsh T, Harrell MI, et al. Germline and somatic mutations in homologous recombination genes predict platinum response and survival in ovarian, fallopian tube, and peritoneal carcinomas. Clin Cancer Res. 2014;20(3):764-775.
- González-Martín A, Harter P, Leary A, et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(10):833-848.
- Capoluongo E, Ellison G, López-Guerrero JA, et al. Guidance statement on BRCA1/2 tumor testing in ovarian cancer patients. Semin Oncol. 2017;44(3):187-197.
- Miller RE, Leary A, Scott CL, et al. ESMO recommendations on predictive biomarker testing for homologous recombination deficiency and PARP inhibitor benefit in ovarian cancer. Ann Oncol. 2020;31(12):1606-1622.
- Gaillard S, Lacchetti C, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: ASCO guideline update. J Clin Oncol. 2025;43(7):868-891.
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer. V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed August 27, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
- Konstantinopoulos PA, Norquist B, Lacchetti C, et al. Germline and somatic tumor testing in epithelial ovarian cancer: ASCO guideline. J Clin Oncol. 2020;38(11):1222-1245.
- Myriad Genetic Laboratories, Inc. Myriad BRACAnalysis CDx® Technical Information. Accessed July 2, 2025. https://myriad-library.s3.amazonaws.com/technical-specifications/BRACAnalysis_CDx_Tech_Specs.pdf
- Myriad Genetic Laboratories, Inc. BRACAnalysis CDx® Germline Companion Diagnostic Test. Accessed August 27, 2025. myriad.com/genetic-tests/bracanalysiscdx-germline-test/
- Myriad Genetic Laboratories, Inc. Myriad MyChoice® CDx Technical Information. Accessed June 30, 2025. https://s3.amazonaws.com/myriad-web/myChoiceCDx/downloads/myChoiceCDxTech.pdf.
- Tan H, Hosein PJ. Detection and therapeutic implications of homologous recombination repair deficiency in pancreatic cancer: A narrative review. J Gastrointest Oncol. 2023;14(05):2249-2259.
- Foundation Medicine. FoundationOne® CDx Technical Information. RAL-0003_v31. Cambridge, MA:2025.
Abbreviations:
BRCAm, BRCA mutation; CDx, companion diagnostic; FFPE, formalin-fixed, paraffin-embedded; gBRCAm, germline BRCA mutation; HRD, homologous recombination deficiency; HRD+, homologous recombination deficiency-positive; HRD–, homologous recombination deficiency-negative; HRR, homologous recombination repair; LOH, loss of heterozygosity; LST, large-scale transitions; NCCN, National Comprehensive Cancer Network® (NCCN®); PARP, poly (ADP-ribose) polymerase; sBRCAm, somatic BRCA mutation; TAI, telomeric allelic imbalance.