Patients with advanced ovarian cancer have a poor prognosis, and most will experience disease recurrence.1-3 Biomarker testing is critical to optimize treatment plans.4,5
Ovarian cancer is the most common cause of death among gynecologic cancers in the US.6
Click to flip
In the US, there were an estimated 20,890 new cases and 12,730 deaths from ovarian cancer in 2025.6
It is the sixth most common cause of cancer deaths among women, representing 4% of all deaths.6
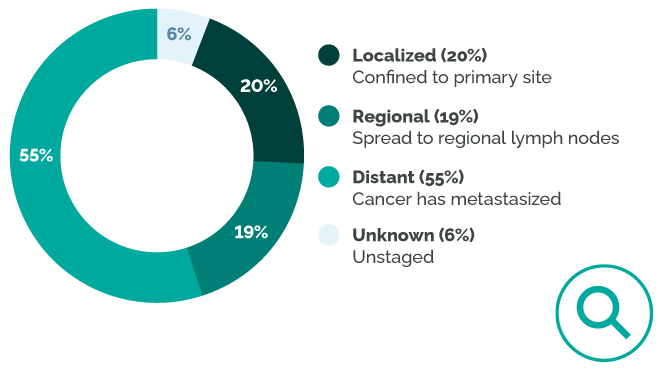
75% of patients are diagnosed with disease that has spread beyond the ovaries.2
Click to flip
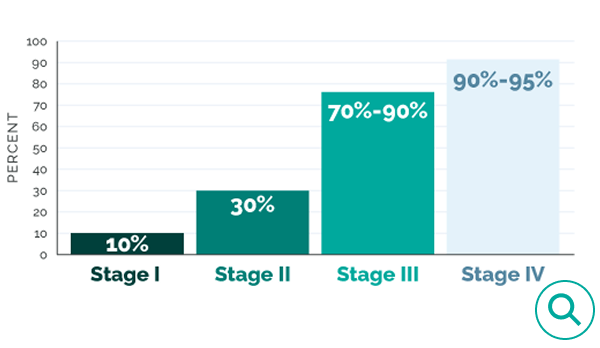
70%–95% of patients with advanced ovarian cancer will experience disease recurrence.1
Click to flip
Women with advanced ovarian cancer are at risk of relapse.9
Click to flip
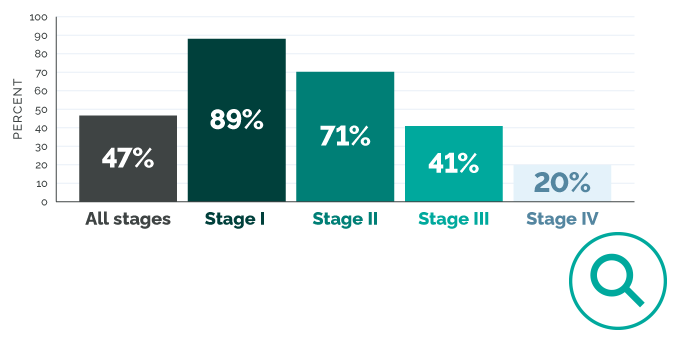
All patients are at risk of recurrence.9
Women with Stage III disease have a ≥70% chance of recurrence.1
Patients with relapsed ovarian cancer tend to experience multiple relapses and undergo multiple rounds of therapy.10
Click to flip
Relapsed ovarian cancer is considered incurable.3,9
Click to flip
Relapsed ovarian cancer is considered incurable and most patients will die from the disease.3,9
Therefore, the first line is likely to be the best setting for treatments to help prevent recurrence.9,15
The treatment goal in some patients with ovarian cancer (particularly those with stage II and III disease) is long-term remission.16
Maintenance treatment following response to first-line platinum-based chemotherapy can impact long-term outcomes for patients with HRD-positive advanced ovarian cancer.17
The first line is likely to be the best setting for treatments to help prevent recurrence.9,15
Maintenance therapy with olaparib plus bevacizumab is an FDA-approved, guideline-recommended treatment option in selected patients who are in complete or partial response to first-line platinum-based chemotherapy.4,5,18,19
LYNPARZA is indicated in combination with bevacizumab for the maintenance treatment of adult patients with advanced epithelial ovarian, fallopian tube or primary peritoneal cancer who are in complete or partial response to first-line platinum-based chemotherapy and whose cancer is associated with HRD-positive status defined by either:
- a deleterious or suspected deleterious BRCA mutation, and/or
- genomic instability
Select patients for therapy based on an FDA-approved companion diagnostic for LYNPARZA.19
Physician Perspectives on Unmet Needs in Advanced Ovarian Cancer

Dr David O’Malley explains why there is no ‘low-risk’ advanced ovarian cancer.
“…if you have 85% chance of recurring, how can you be low risk?”

Dr John Chan emphasizes that all women are at risk of recurrence and the goal of maintenance therapy is to increase the treatment-free interval.
“All women with stage III or IV ovarian cancer are at high risk of recurrence”
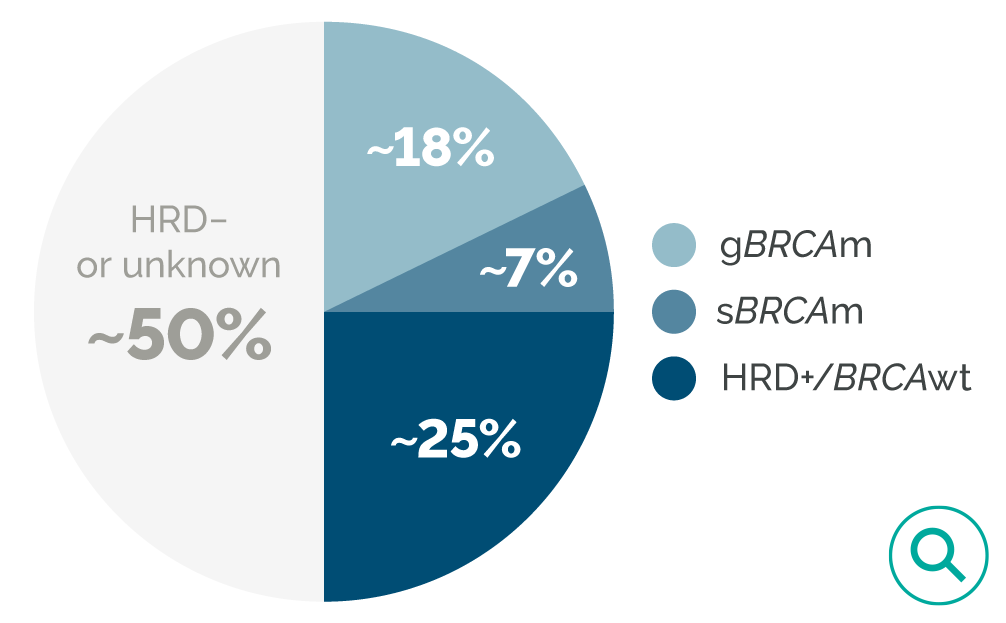
HRD testing is critical to inform eligibility of patients with advanced ovarian cancer for the PAOLA-1 treatment regimen.19
HRD is associated with increased sensitivity to PARP inhibitor maintenance therapy.22
The FDA-approved indication of LYNPARZA plus bevacizumab requires a positive HRD test with an FDA-approved companion diagnostic.19,23
Treatment guidelines recommend genetic biomarker testing in all patients.4,24
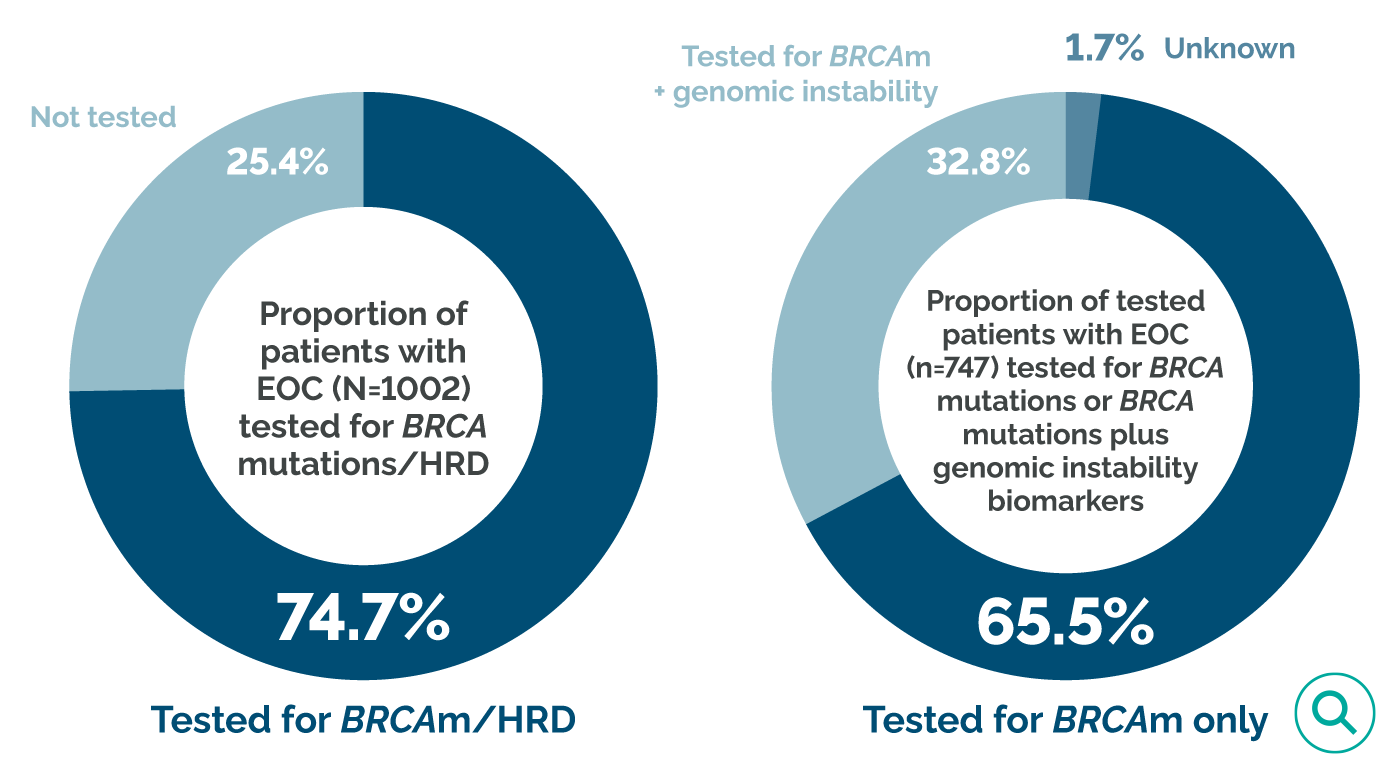
Many patients with ovarian cancer are not tested for BRCA mutations or HRD, so their eligibility for certain targeted therapies is not known.19,25
In a retrospective study of biomarker testing in 1002 patients with epithelial ovarian cancer drawn from community health systems in the US (2018–2020), 74.7% (748/1002) underwent testing, with 1 patient having an invalid test result.25
Of the 747 patients with a valid test result25:
- 65.5% (489/747) were tested for BRCA mutations only
- Only 32.8% (245/747) were tested for BRCA mutations and measures of genomic instability beyond BRCA (eg, LOH, GIS)
Testing for HRD is required to identify patients eligible for LYNPARZA plus bevacizumab.19
In advanced ovarian cancer, targeted first-line maintenance therapy provides clinical benefit by prolonging disease control.26
The combination of LYNPARZA plus bevacizumab as first-line maintenance therapy in advanced ovarian cancer was examined in the PAOLA-1 clinical trial.15
PAOLA-1 resulted in an approval for use in selected patients with HRD-positive* advanced ovarian cancer, based on an FDA-approved test.19
Treatment guidelines recommend the combination of olaparib (LYNPARZA) plus bevacizumab as first-line maintenance therapy option in selected patients with advanced ovarian cancer in complete or partial response to first-line platinum-based therapy, depending on biomarker status.4,5,18
*Including BRCA mutation (as determined by Myriad MyChoice® CDx) and other causes of HRD. HRD-positive is defined as either a tumor BRCA mutation and/or an HRD score ≥42 by Myriad MyChoice® CDx.23,27

Dr David O’Malley discusses educating patients about maintenance therapy.
“It is no longer ‘should we be giving maintenance therapy’… it is ‘which maintenance therapy should we be giving’.”
Why use combination maintenance therapy in patients with advanced ovarian cancer?
The PARP inhibitor LYNPARZA® (olaparib) is used as monotherapy and in combination with the angiogenesis inhibitor bevacizumab as maintenance therapy in advanced ovarian cancer.19,28
Cancer cells may rely on distinct and complementary mechanisms for survival.29 Combination therapy with LYNPARZA plus bevacizumab was designed to target 2 mechanisms commonly seen in ovarian tumors.20,29,30

To learn about the mechanism of action* of these drugs, the rationale for using them as combination therapy, and a population-adjusted indirect treatment comparison (ITC) of the BRCA-mutated populations of the SOLO-1 (LYNPARZA) and PAOLA-1 (LYNPARZA plus bevacizumab) trials review the following slide deck and infographic.
*The exact mechanism of disease in the context of combination treatment strategies is yet to be elucidated, and the exact mechanism of action of LYNPARZA is unknown.

Dr Ramez Iskander explains the ITC analysis of the SOLO-1 and PAOLA-1 results.
“This analysis… was completed because currently there are no randomized controlled trials that investigate maintenance PARP inhibitor monotherapy, bevacizumab monotherapy, PARP inhibitor plus bevacizumab combination therapy, and watch-and-wait placebo.”
The design, efficacy, and safety results of the SOLO-1 and PAOLA-1 trials are described in detail on lynparzahcp.com.
References
- Ovarian Cancer Research Alliance. Recurrence. Accessed August 27, 2025. https://ocrahope.org/patients/about-ovarian-cancer/recurrence/
- National Cancer Institute (NCI). SEER Cancer Stat Facts: Ovarian Cancer. Bethesda, MD: 2025. Accessed August 27, 2025. https://seer.cancer.gov/statfacts/html/ovary.html
- Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379(26):2495-2505.
- González-Martín A, Harter P, Leary A, et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(10):833-848.
- Gaillard S, Lacchetti C, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: ASCO guideline update. J Clin Oncol. 2025;43(7):868-891.
- American Cancer Society (ACS). Cancer Facts & Figures 2025. Atlanta, GA; 2025. Accessed August 28, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2025/2025-cancer-facts-and-figures-acs.pdf
- Torre LA, Trabert B, DeSantis CE, et al. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018;68(4):284-296.
- Caruso G, Tomao F, Parma G, et al. Poly (ADP-ribose) polymerase inhibitors (PARPi) in ovarian cancer: Lessons learned and future directions. Int J Gynecol Cancer. 2023;33(4):431-443.
- Lorusso D, Mouret-Reynier M-A, Harter P, et al. Updated progression-free survival and final overall survival with maintenance olaparib plus bevacizumab according to clinical risk in patients with newly diagnosed advanced ovarian cancer in the phase III PAOLA-1/ENGOT-ov25 trial. Int J Gynecol Cancer. 2024;34(4):550-558.
- Bolívar PS, Gonzalez-Benitez C, Carbonell López MC, et al. Prognostic factors after the first recurrence of ovarian cancer. J Clin Med. 2025;14(2):470.
- Eng KH, Hanlon BM, Bradley WH, Szender JB. Prognostic factors modifying the treatment-free interval in recurrent ovarian cancer. Gynecol Oncol. 2015;139(2):228-235.
- Gupta S, Nag S, Aggarwal S, Rauthan A, Warrier N. Maintenance therapy for recurrent epithelial ovarian cancer: Current therapies and future perspectives – a review. J Ovarian Res. 2019;12(1):103.
- Dunton CJ. Management of treatment-related toxicity in advanced ovarian cancer. Oncologist. 2002;7(Suppl 5):11-19.
- Lutgendorf SK, Shinn E, Carter J, et al. Quality of life among long-term survivors of advanced stage ovarian cancer: A cross-sectional approach. Gynecol Oncol. 2017;146(1):101-108.
- Ray-Coquard I, Leary A, Pignata S, et al. Olaparib plus bevacizumab first-line maintenance in ovarian cancer: final overall survival results from the PAOLA-1/ENGOT-ov25 trial. Ann Oncol. 2023;34(8):681-692.
- Coleman RL, Segunmaru Z, Simmons D, et al. Oncologists’ perspectives on discussing ‘cure’ with patients with ovarian cancer. Poster presented at: SGO 2024 Annual Meeting on Women’s Cancer; March 16-18, 2024; San Diego, CA.
- Chan JK, Liu J, Song J, et al. Real-world outcomes associated with poly (ADP-ribose) polymerase inhibitor monotherapy maintenance in patients with primary advanced ovarian cancer. Am J Clin Oncol. 2023;46(7):314-322.
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Ovarian Cancer, Including Fallopian Tube Cancer and Primary Peritoneal Cancer. V.3.2025. ©National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed August 27, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
- LYNPARZA® (olaparib) [Prescribing Information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025.
- Konstantinopoulos PA, Ceccaldi R, Shapiro GI, D’Andrea AD. Homologous recombination deficiency: exploiting the fundamental vulnerability of ovarian cancer. Cancer Discov. 2015;5(11):1137-1154.
- Pennington KP, Walsh T, Harrell MI, et al. Germline and somatic mutations in homologous recombination genes predict platinum response and survival in ovarian, fallopian tube, and peritoneal carcinomas. Clin Cancer Res. 2014;20(3):764-775.
- Ngoi NYL, Tan DSP. The role of homologous recombination deficiency testing in ovarian cancer and its clinical implications: do we need it? ESMO Open. 2021;6(3):100144.
- Myriad Genetic Laboratories, Inc. Myriad MyChoice® CDx Technical Information. Accessed June 30, 2025. https://s3.amazonaws.com/myriad-web/myChoiceCDx/downloads/myChoiceCDxTech.pdf.
- Konstantinopoulos PA, Norquist B, Lacchetti C, et al. Germline and somatic tumor testing in epithelial ovarian cancer: ASCO guideline. J Clin Oncol. 2020;38(11):1222-1245.
- Matsuno RK, Williams AFO, Sweetnam C, et al. Patterns of homologous repair deficiency and BRCA1/2 testing of ovarian and breast cancers: A real-world study of patients in community health settings in the United States. JCO Oncol Adv. 2025;2(1).
- Elyashiv O, Wong YN, Ledermann JA. Frontline maintenance treatment for ovarian cancer. Curr Oncol Rep. 2021;23(8):97.
- Ray-Coquard I, Pautier P, Pignata S, et al; PAOLA-1 Investigators . Olaparib plus bevacizumab as first-line maintenance in ovarian cancer. N Engl J Med. 2019;381(25):2416-2428.
- Avastin® (bevacizumab) [prescribing information]. South San Francisco, CA: Genentech, Inc; 2022.
- Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144(5):646-674.
- Moghaddam SM, Amini A, Morris DL, Pourgholami MH. Significance of vascular endothelial growth factor in growth and peritoneal dissemination of ovarian cancer. Cancer Metastasis Rev. 2012;31(1-2):143-162.
Abbreviations:
BRCAwt, BRCA wild-type; CDx, companion diagnostic; CESD, Center for Epidemiological Studies Depression; FACT-O, Functional Assessment of Cancer Therapy – Ovarian; FDA, US Food and Drug Administration; gBRCAm, germline BRCA mutation; GIS, genomic instability score; HRD, homologous recombination deficiency; HRD+, homologous recombination deficiency-positive; HRD–, homologous recombination deficiency–negative; IPAQ-SF, International Physical Activity Questionnaire-Short Form; ITC, indirect treatment comparison; PARP, poly (ADP-ribose) polymerase; QoL, quality of life; sBRCAm, somatic BRCA mutation; SPS, Social Provisions Scale.