HRD Testing and Treatment Guidelines in Advanced Ovarian Cancer
Key treatment guidelines recommend HRD and BRCA mutation testing in patients with advanced ovarian cancer. Testing should be performed at diagnosis.1-5
Select buttons to learn more about HRD and BRCA mutation testing guidelines.
Whom to Test?
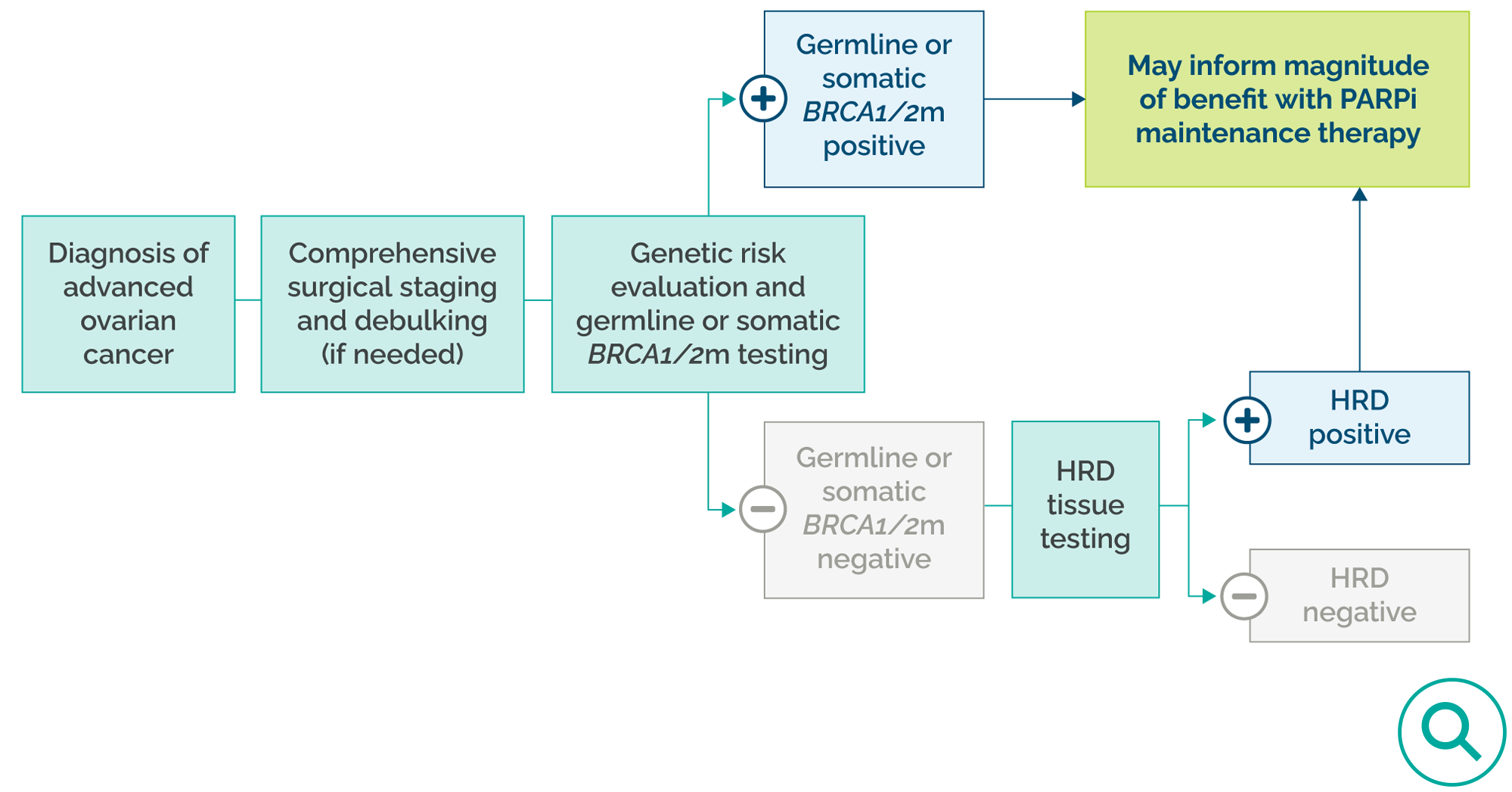
All patients with ovarian cancer, fallopian tube cancer, or primary peritoneal cancer should have genetic risk evaluation and germline and somatic testing (if not previously done).1
What to Test?
Germline testing: BRCA1 and BRCA2 status should be tested.1
Tumor testing: In the up-front setting, choice of somatic testing should, at a minimum, optimize identification of molecular alterations that can inform use of interventions that have demonstrated benefit in this setting, including BRCA1 and BRCA2, loss of heterozygosity, or HRD status in the absence of a germline BRCA mutation.1
In the absence of BRCA1/2 status, HRD status may provide information on the magnitude of benefit of PARP inhibitor therapy.1
When to Test?
Upon pathologic confirmation of ovarian cancer, fallopian tube cancer, or primary peritoneal cancer, patients should be referred for a genetic risk evaluation and germline and somatic testing (if not previously done).1
NCCN ovarian cancer biomarker testing recommendations1
BRCA1/2m, BRCA1 or BRCA2 mutation; HRD, homologous recombination deficiency; PARPi, poly (ADP-ribose) polymerase inhibitor.
Whom to Test?
All women diagnosed with epithelial ovarian cancer should be offered germline genetic testing irrespective of their clinical features or family cancer history. Somatic tumor testing should be performed in women, who do not carry a germline pathogenic or likely pathogenic BRCA1/2 variant3
What to Test?
Germline: BRCA1, BRCA2, and other ovarian cancer susceptibility genes3
Somatic: Somatic tumor testing for BRCA1 and BRCA2 pathogenic or likely pathogenic variants3
HRD: Somatic tumor testing should involve measure(s) of homologous recombination4
When to Test?
Testing should occur at the time of diagnosis or as soon as feasibly possible4
Whom to Test?
The SGO recommends universal germline genetic assessment for all those diagnosed with epithelial ovarian cancer.5
What to Test?
For patients undergoing upfront treatment for ovarian cancer with negative germline genetic testing, somatic tumor testing should, at a minimum, include BRCA1/2 somatic variants, LOH, and HRD status as these findings can guide the use of PARP inhibitor maintenance therapy.5
When to Test?
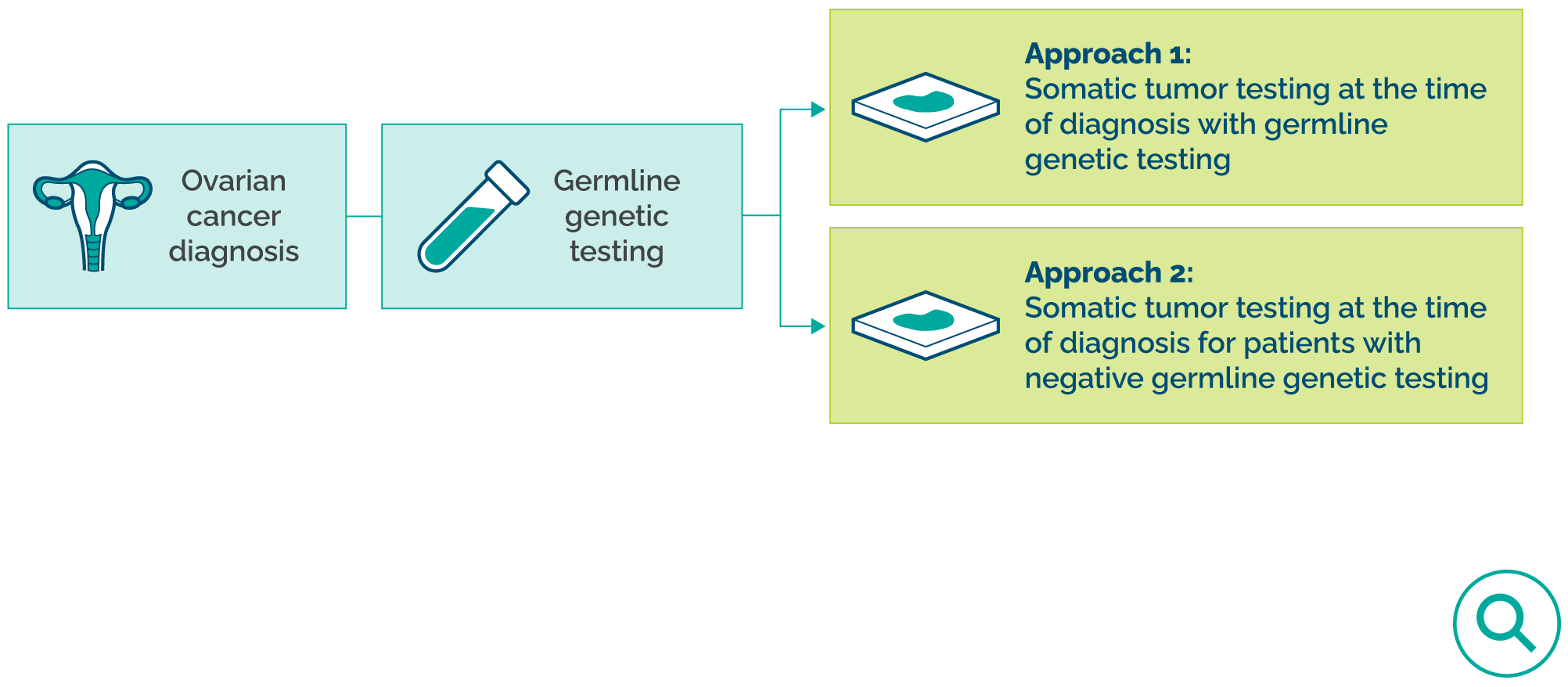
Test all patients at the time of disease diagnosis; with 2 possible approaches5
-
- Approach 1: Order somatic tumor testing for HRD with germline BRCA testing
- Approach 2: Order somatic tumor testing for HRD for patients with negative germline BRCA testing
| Guideline | Whom to test | What to test | When to test |
|---|---|---|---|
| GuidelineNCCN |
|
|
When to testUpon pathologic confirmation of OC, FTC, or PPC |
| GuidelineASCO |
|
|
When to testAt diagnosis or as soon as is feasibly possible |
| GuidelineSGO |
|
|
When to testAt diagnosis |
ASCO, American Society of Clinical Oncology; FTC, fallopian tube cancer; HRD, homologous recombination deficiency; LOH, loss of heterozygosity; NCCN, National Comprehensive Cancer Network; OC, ovarian cancer; PPC, primary peritoneal cancer; SGO, Society of Gynecologic Oncology.
NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
Treatment guidelines recommend olaparib in combination with bevacizumab1,2,4,6
Treatment guidelines recommend the combination of olaparib plus bevacizumab as first-line maintenance therapy as a treatment option in selected patients with advanced ovarian cancer in complete or partial response to first-line therapy, depending on biomarker status.1,2,4,6
| BRCAm | HRD+/BRCAwt | Notes | |
|---|---|---|---|
| NCCN1 | BRCAm |
HRD+/BRCAwt |
NotesNCCN Category 1* recommendation, only in patients that received bevacizumab as part of primary therapy. |
| ASCO4,6 | BRCAm |
HRD+/BRCAwt |
Notes |
Olaparib in combination with bevacizumab is the ONLY PARP inhibitor with an NCCN Category 1* recommendation as a treatment option for first-line maintenance treatment in HRD-positive advanced ovarian cancer in patients who received bevacizumab as part of primary therapy.1
NCCN Guidelines recommendations for olaparib plus bevacizumab as post-primary maintenance therapy
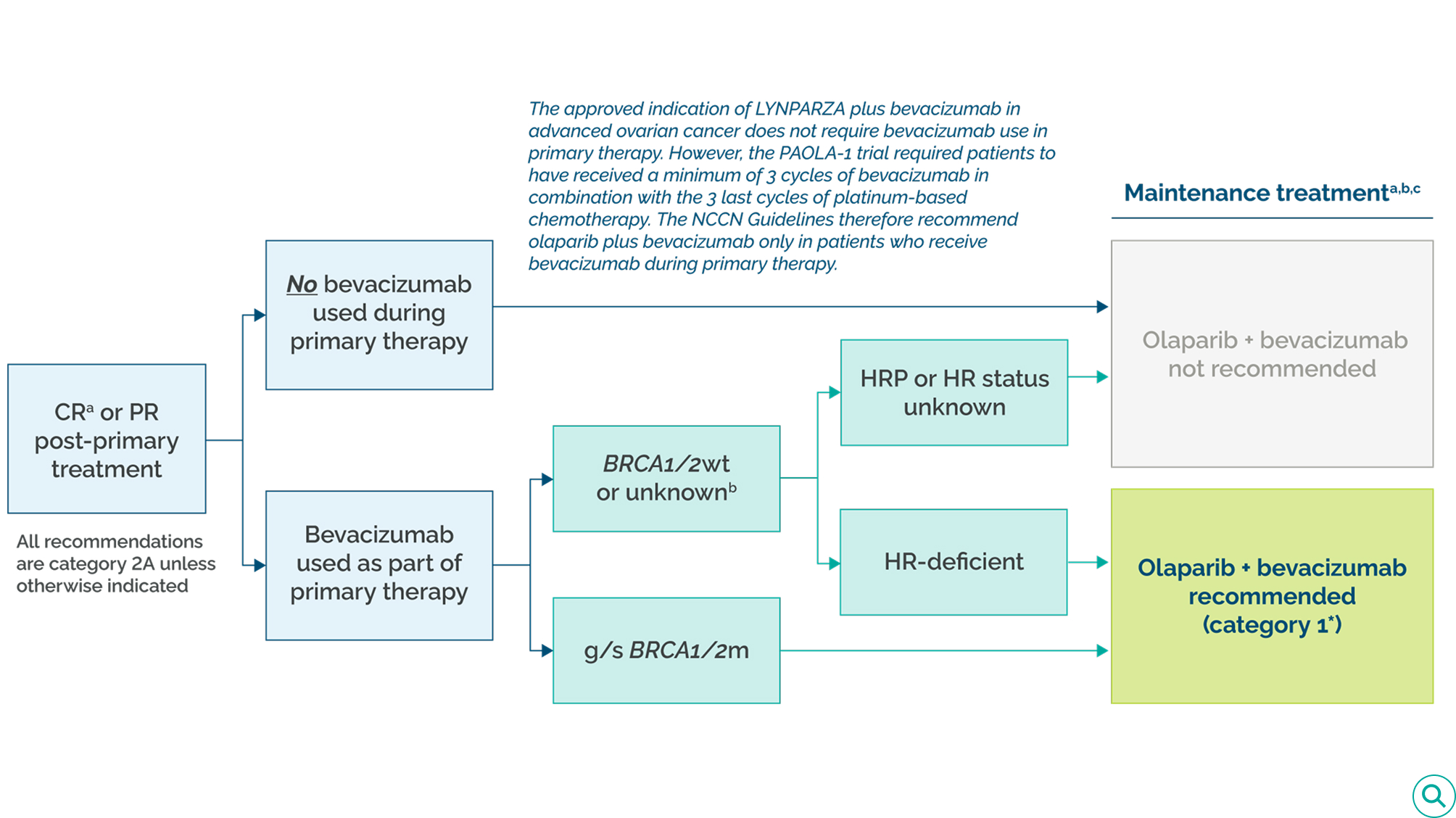
aNo definitive evidence of disease.
bIn the absence of a BRCA1/2 mutation, HRD status may provide information on the magnitude of benefit of PARP inhibitor therapy.
cSee Principles of Systemic Therapy (OV-C) and Management of Drug Reactions (OV-D), NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Ovarian Cancer (including Fallopian Tube Cancer and Primary Peritoneal Cancer) V.3.2025.
*NCCN Category 1: Based upon high-level evidence (≥1 randomized phase 3 trials or high-quality, robust meta-analyses), there is uniform NCCN consensus (≥85% support of the Panel) that the intervention is appropriate.
Adapted with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Ovarian Cancer/Fallopian Tube Cancer/Primary Peritoneal Cancer V.3.2025. © 2025 National Comprehensive Cancer Network, Inc. All rights reserved. The NCCN Guidelines® and illustrations herein may not be reproduced in any form for any purpose without the express written permission of NCCN. To view the most recent and complete version of the NCCN Guidelines, go online to NCCN.org. The NCCN Guidelines are a work in progress that may be refined as often as new significant data becomes available. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
References
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer. V.3.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed August 27, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way
- González-Martín A, Harter P, Leary A, et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(10):833-848.
- Konstantinopoulos PA, Norquist B, Lacchetti C, et al. Germline and somatic tumor testing in epithelial ovarian cancer: ASCO guideline. J Clin Oncol. 2020;38(11):1222-1245.
- Gaillard S, Lacchetti C, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: ASCO guideline update. J Clin Oncol. 2025;43(7):868-891.
- Gressel GM, Frey MK, Norquist B, Senter L, Blank SV, Urban RR. Germline and somatic testing for ovarian cancer: An SGO clinical practice statement. Gynecol Oncol. 2024;181:170-178.
- Tew WP, Lacchetti C, Kohn EC. PARP inhibitors in the management of ovarian cancer: ASCO guideline rapid recommendation update. J Clin Oncol. 2022;40(33):3878-3881.
Abbreviations:
ASCO, American Society of Clinical Oncology; BRCAm, BRCA mutation; BRCAwt, BRCA wildtype; ESMO, European Society for Medical Oncology; HRD, homologous recombination deficiency; HRD+, homologous recombination deficiency-positive; HRD–, homologous recombination deficiency-negative; NCCN, National Comprehensive Cancer Network® (NCCN®); PARP, poly (ADP ribose) polymerase; PARPi, poly (ADP ribose) polymerase inhibitor; SGO, Society of Gynecologic Oncology.