BRCA and homologous recombination deficiency (HRD) testing are required to determine eligibility for LYNPARZA monotherapy and LYNPARZA plus bevacizumab combination therapy.1
LYNPARZA® (olaparib) plus bevacizumab is a treatment option for selected patients with HRD-positive advanced ovarian cancer.1
LYNPARZA is approved both as monotherapy and in combination with bevacizumab as first-line maintenance therapy in selected patients with advanced ovarian cancer and both indications require a diagnostic test.
- The results of the SOLO-1 clinical trial of LYNPARZA supported monotherapy as a treatment for selected patients with BRCA-mutated advanced ovarian cancer1
- The results of the PAOLA-1 clinical trial of LYNPARZA plus bevacizumab supported the combination as a treatment for selected patients with HRD-positive advanced ovarian cancer1
According to drug indications and treatment guidelines:
- Select patients with BRCA mutations are eligible for LYNPARZA monotherapy1-3
- Select patients with HRD-positive disease are eligible for LYNPARZA plus bevacizumab1-3
- HRD-positive is defined as either a tumor BRCA mutation and/or an HRD score of ≥42 by Myriad MyChoice® CDx.4,5
Click on the buttons below to see details of the two trials.
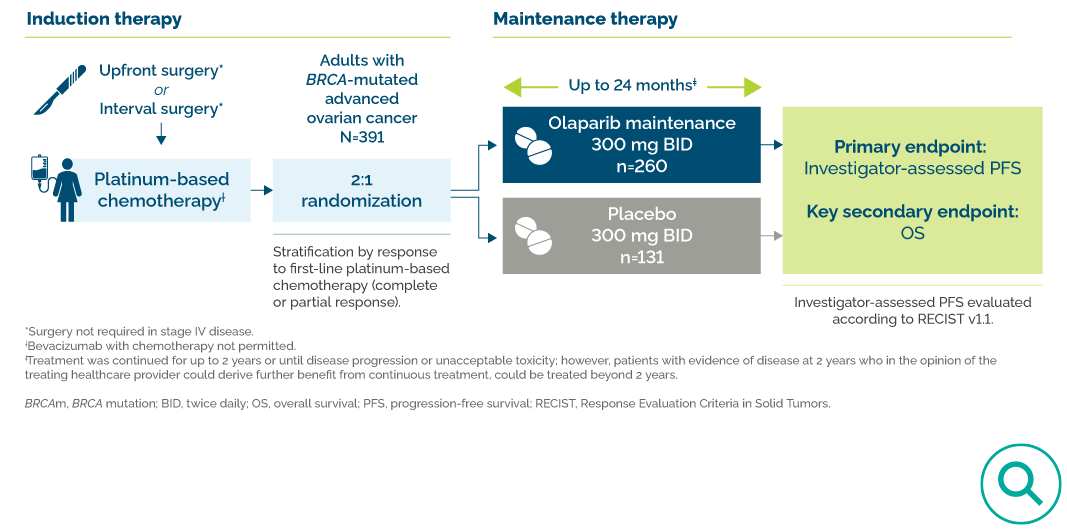
In SOLO-16:
- All patients had a germline or somatic BRCA1 or BRCA2 mutation
- Patients with stage III disease underwent surgery, either before (upfront) or during (interval) chemotherapy; patients with stage IV disease may not have undergone surgery
- Intravenous (IV) platinum-based chemotherapy
- No prior bevacizumab use
- LYNPARZA, after chemotherapy, for up to 24 months total6
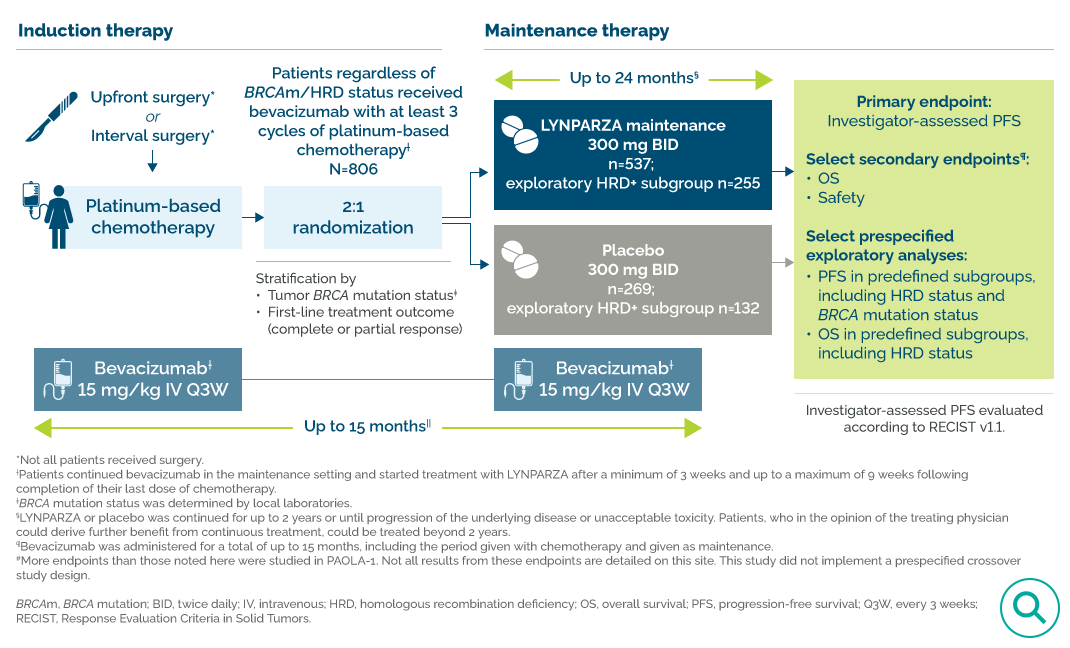
In PAOLA-15:
- Patients were eligible regardless of BRCA mutation or HRD status5
- Some patients underwent surgery, either before (upfront) or during (interval) chemotherapy5
- Intravenous (IV) platinum plus taxane chemotherapy5
- IV bevacizumab, during and after chemotherapy, for 15 months total5
- Olaparib, after chemotherapy, for 24 months total5
Treatment Selection – Patient Cases
The patient cases presented below provide examples of biomarker testing and treatment selection in hypothetical patients, identifying those eligible for LYNPARZA plus bevacizumab.
PAOLA-1 Trial Design
PAOLA-1 studied LYNPARZA plus bevacizumab vs an active comparator (bevacizumab plus placebo) as maintenance therapy in HRD-positive advanced ovarian cancer after response to first-line platinum-based chemotherapy.5
PAOLA-1 was the first phase 3 trial to investigate a PARP inhibitor combination regimen against an established maintenance therapy—bevacizumab.5

Dr Thomas J Herzog discusses the design of the PAOLA-1 trial and population.
“The treatment effect of the combination regimen was compared to an active bevacizumab control arm, not watch-and-wait placebo.”

Dr Ramez Eskander explains the lack of an olaparib monotherapy arm in the PAOLA-1 trial.
“At the time PAOLA-1 was designed, we didn’t know the SOLO-1 outcome”
PAOLA-1 Efficacy Results
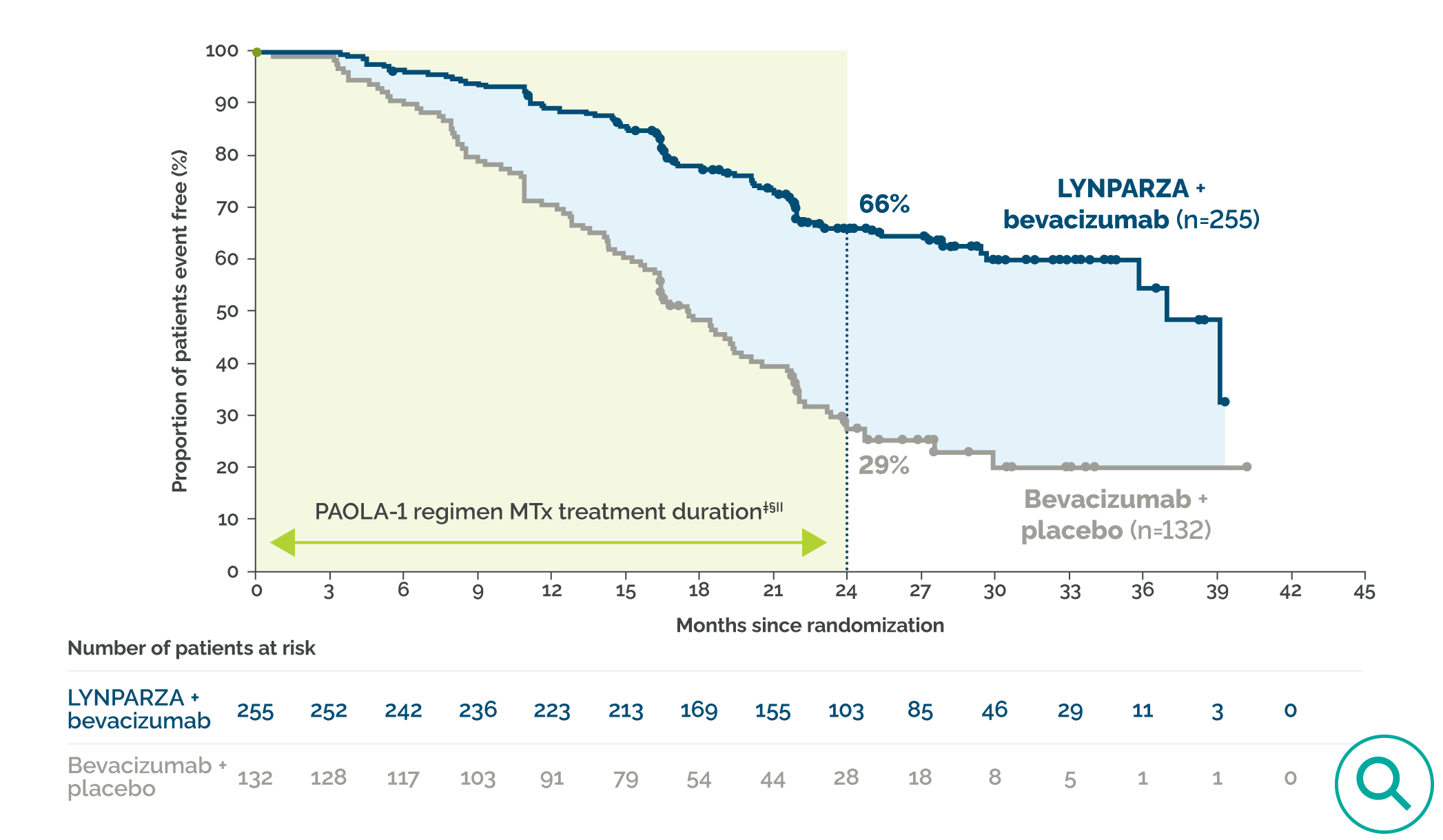
Prespecified Exploratory Endpoint: PFS in the HRD-Positive Subgroup
-
- FDA approval was based on a prespecified exploratory HRD-positive* subgroup.1,5
- A prespecified exploratory subgroup analysis showed clinically meaningful PFS benefit in HRD-positive (including tumor BRCA-mutated) patients after response to first-line platinum-based chemotherapy.1,5,7†
- Median PFS was 3.1 years (37.2 months) with LYNPARZA plus bevacizumab vs ~1.5 years (17.7 months) with bevacizumab plus placebo5
- 67% risk reduction of disease progression or death; HR=0.33 (95% CI: 0.25–0.45)5
- Data was based upon a prespecified exploratory subgroup analysis, which was not controlled for Type 1 error. HRD status was not a stratification factor in PAOLA-1.8
- In a prespecified exploratory analysis of HRD-negative and HRD-unknown patients, there was insufficient evidence to suggest differential efficacy between the combination of LYNPARZA plus bevacizumab and bevacizumab plus placebo.5
§LYNPARZA was continued for up to 2 years or until progression of the underlying disease or unacceptable toxicity. Patients, who in the opinion of the treating physician could derive further benefit from continuous treatment, could be treated beyond 2 years.1 ||Patients with a complete response should stop treatment at 2 years. Patients with evidence of disease at 2 years can remain on therapy at physician discretion.1 In PAOLA-1, it was unknown how many HRD-positive patients remained on therapy longer than 2 years; therefore, results should be interpreted with caution.
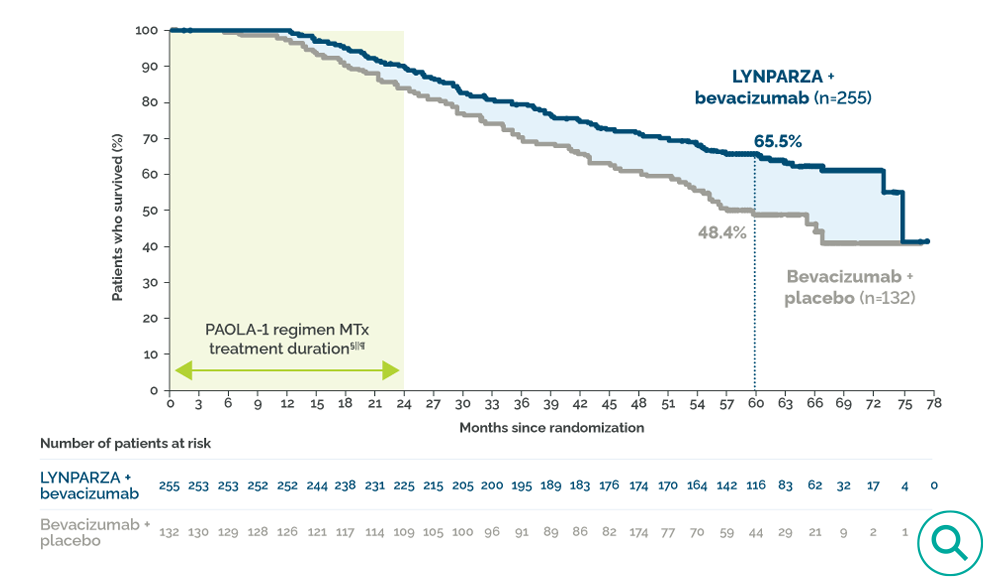
Prespecified Exploratory Endpoint: OS in the HRD-Positive Subgroup
-
- Prespecified exploratory analysis of the secondary endpoint overall survival (OS) in the HRD-positive*† subgroup showed a clinically meaningful survival benefit after response to first-line platinum-based chemotherapy.7,8‡
- Median OS was ~6.3 years (75.2 months) with LYNPARZA plus bevacizumab vs ~4.8 years (57.3 months) with bevacizumab plus placebo8
- 38% reduction in the risk of death; HR=0.62 (95% CI: 0.45–0.85)
- Data based upon a prespecified exploratory subgroup analysis, which was not controlled for Type 1 error. HRD status was not a stratification factor in PAOLA-1. The analysis is based on Kaplan–Meier estimates and is descriptive only. This trial was not designed to assess a statistical difference between treatment groups at 5 years.8
LYNPARZA in combination with bevacizumab is the only maintenance combination therapy to demonstrate a clinically meaningful improvement in OS outcomes in patients with HRD-positive disease after response to first-line platinum-based chemotherapy.†
†Including BRCA mutation (as determined by Myriad MyChoice® CDx) and other causes of HRD. HRD-positive is defined as either a tumor BRCA mutation and/or an HRD score ≥42 by Myriad MyChoice® CDx.4,5
‡Secondary endpoint: Prespecified exploratory analysis of OS in the HRD-positive subgroup. Data based upon a prespecified exploratory subgroup analysis, which was not controlled for Type 1 error. HRD status was not a stratification factor in PAOLA-1. The analysis is based on Kaplan–Meier estimates and is descriptive only. This trial was not designed to assess a statistical difference between treatment groups at 5 years.8
§Bevacizumab was administered for a total of up to 15 months, including the period given with chemotherapy and given as maintenance.5
||LYNPARZA was continued for up to 2 years or until progression of the underlying disease or unacceptable toxicity. Patients, who in the opinion of the treating physician could derive further benefit from continuous treatment, could be treated beyond 2 years.1
¶Patients with a complete response should stop treatment at 2 years. Patients with evidence of disease at 2 years can remain on therapy at physician discretion.1 In PAOLA-1, it was unknown how many HRD-positive patients remained on therapy longer than 2 years; therefore, results should be interpreted with caution.

Dr Ramez Eskander outlines the key efficacy results from the PAOLA-1 trial.
“This is the first and only phase three clinical trial to investigate a PARP inhibitor as a combination regimen… against an active comparator, an established maintenance therapy of bevacizumab.”
PAOLA-1 Safety Results
Common adverse reactions (ARs) with LYNPARZA were generally consistent with the known safety profile of LYNPARZA monotherapy.1
ARs and laboratory abnormalities from the primary analysis in PAOLA-1 were mostly Grades 1 and 2.1
8 out of 10 patients remained on LYNPARZA as prescribed, in combination with bevacizumab, without discontinuing due to ARs.1
Primary analysis:
Fatal adverse reactions occurred in 1 patient due to concurrent pneumonia and aplastic anemia. Serious adverse reactions occurred in 31% of patients who received LYNPARZA plus bevacizumab. Serious adverse reactions in >5% of patients included hypertension (19%) and anemia (17%).1
In addition, venous thromboembolism occurred more commonly in patients receiving LYNPARZA plus bevacizumab (5%) than in those receiving placebo plus bevacizumab (1.9%)1
Primary analysis: ARs occurring in ≥10% of patients treated with LYNPARZA plus bevacizumab and ≥5% frequency compared with placebo plus bevacizumab1
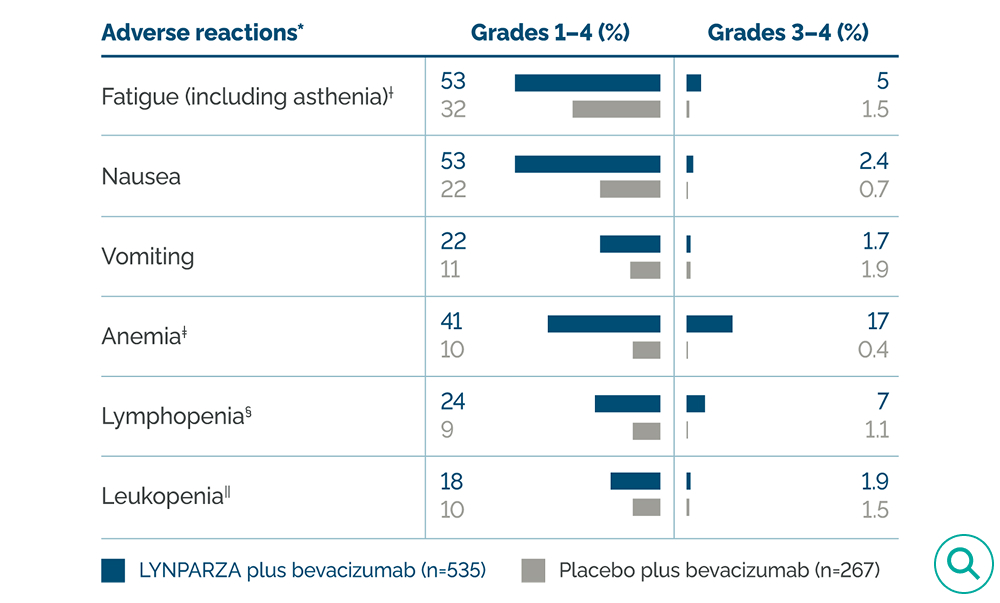
†Includes asthenia and fatigue.
‡Includes anemia, anemia macrocytic, erythropenia, hematocrit decreased, hemoglobin decreased, normochromic anemia, normochromic normocytic anemia, normocytic anemia, and red blood cell count decreased.
§Includes B-lymphocyte count decreased, lymphocyte count decreased, lymphopenia, and T-lymphocyte count decreased.
||Includes leukopenia and white blood cell count decreased.
At 5-year follow-up analysis:
- No new safety signals were identified8
- The incidence of MDS/AML/AA was 1.7% (9/535) in the LYNPARZA plus bevacizumab group and 2.2% (6/267) in the bevacizumab plus placebo group8
- In the HRD-positive subgroup, the incidence of MDS/AML was 1.6% (4/255) in patients who received LYNPARZA plus bevacizumab and 2.3% (3/131) in patients who received bevacizumab plus placebo1
- 22 (4.1%) new primary malignancy events occurred in the LYNPARZA plus bevacizumab group and 8 (3.0%) events occurred in the bevacizumab plus placebo group8
- 7 (1.3%) pneumonitis events occurred in the LYNPARZA plus bevacizumab group and 2 (0.7%) events occurred in the bevacizumab plus placebo group8

Dr Herzog summarizes the safety results of PAOLA-1.
“The AE profile of LYNPARZA in PAOLA-1 was consistent with previous trials of LYNPARZA”

Dr Ramez Eskander describes safety data from the PAOLA-1 trial, including key adverse reactions (ARs) in patients receiving olaparib.
“This slide shows the adverse events that occurred in greater or equal to 10% of patients that were receiving both olaparib plus bevacizumab in the PAOLA-1 clinical trial.”

Kay Harger, RN, describes the management of nausea and fatigue in patients receiving olaparib.
“How do you help us manage these adverse events?”

Dr John Chan explains his considerations when choosing LYNPARZA monotherapy vs LYNPARZA plus bevacizumab.
“In patients who started on chemotherapy plus bevacizumab, who have a BRCA mutation… Do you stop bevacizumab, or continue bevacizumab and give them olaparib plus bevacizumab?”
The design, efficacy, and safety results of the SOLO-1 and PAOLA-1 trials are described in detail on lynparzahcp.com.
References
- LYNPARZA® (olaparib) [prescribing information]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; 2025.
- González-Martín A, Harter P, Leary A, et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(10):833-848.
- Gaillard S, Lacchetti C, Armstrong DK, et al. Neoadjuvant chemotherapy for newly diagnosed, advanced ovarian cancer: ASCO guideline update. J Clin Oncol. 2025;43(7):868-891.
- Myriad Genetic Laboratories, Inc. Myriad MyChoice® CDx Technical Information. Accessed June 30, 2025. https://s3.amazonaws.com/myriad-web/myChoiceCDx/downloads/myChoiceCDxTech.pdf.
- Ray-Coquard I, Pautier P, Pignata S, et al; PAOLA-1 Investigators. Olaparib plus bevacizumab as first-line maintenance in ovarian cancer. N Engl J Med. 2019;381(25):2416-2428.
- Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379(26):2495-2505.
- Ellis LM, Bernstein DS, Voest EE, et al. American Society of Clinical Oncology Perspective: Raising the bar for clinical trials by defining clinically meaningful outcomes. J Clin Oncol. 2014;32(12):1277-1280.
- Ray-Coquard I, Leary A, Pignata S, et al. Olaparib plus bevacizumab first-line maintenance in ovarian cancer: final overall survival results from the PAOLA-1/ENGOT-ov25 trial. Ann Oncol. 2023;34(8):681-692.
- Hardesty MM, Krivak TC, Wright GS, et al. OVARIO phase II trial of combination niraparib plus bevacizumab maintenance therapy in advanced ovarian cancer following first-line platinum-based chemotherapy with bevacizumab. Gynecol Oncol. 2022;166(2):219-229.
- Monk BJ, Oaknin A, O’Malley D et al. ATHENA-COMBO, a phase III, randomized trial comparing rucaparib (RUCA) + nivolumab (NIVO) combination therapy vs RUCA monotherapy as maintenance treatment in patients (pts) with newly diagnosed ovarian cancer (OC). Ann Oncol. 2024;35:1223-1224.
- Ray-Coquard I, Pautier P, Pignata S, et al. Phase III PAOLA-1/ENGOT-ov25: maintenance olaparib with bevacizumab in patients with newly diagnosed, advanced ovarian cancer treated with platinum-based chemotherapy and bevacizumab as standard of care. Presentation LBA2_PR presented at ESMO Annual Conference 2019, 27 September–1 October, Barcelona, Spain.
- Ray-Coquard I, Pautier P, Pignata S, et al. Supplementary Information. Olaparib plus bevacizumab as first-line maintenance in ovarian cancer. N Engl J Med. 2019;381(25):2416-2428.
Abbreviations:
AE, adverse event; aOC, advanced ovarian cancer; AR, adverse reaction; Bev, bevacizumab; BRCAm, BRCA mutation; BRCAwt, BRCA wildtype; BID, twice daily; CDx, companion diagnostic; CI, confidence interval; eCRF, electronic case report form; gBRCAm, germline BRCA mutation; HR, hazard ratio; HRD, homologous recombination deficiency; ITC, indirect treatment comparison; ITT, intent-to-treat; IV, intravenous; NCI CTCAE, National Cancer Institute Common Terminology Criteria for Adverse Events; OS, overall survival; PARP, poly (ADP-ribose) polymerase; PFS, progression-free survival; PO, orally; RECIST, Response Evaluation Criteria In Solid Tumors; Q3W, every 3 weeks; QoL, quality of life; UTI, urinary tract infection.